  
This is because there is carbon dioxide (CO 2) exchange in the atmosphere, which leads to constant turnover of carbon molecules within the body cells. Let's look further at the technique behind the work that led to Libby being awarded a Nobel prize in 1960.Ĭarbon 14 (C-14) is a radioactive element that is found naturally, and a living organism will absorb C-14 and maintain a certain level of it in the body. We started the first article by talking about carbon dating and the Dead Sea scrolls. Similarly, in a population which growsĮxponentially with time there is the concept of "doubling time". The equivalent thickness for the medium in radiation attenuation is known as "half-value thickness". 
Incidentally, our formula for t gives us an easy way of finding the half-life, the time it takes for half the nuclei in a sample to decay. To find, the mean value of time of survival, all we have to do is find the integral Plotting t against F with a value of l=1 gives the graph on the right. Note that that the domain of F is the interval from zero to 1, which corresponds to the interval of time from zero to infinity. Once we have an expression for t, a "definite integral" will give us the mean value of t (this is how "mean value" is defined).įrom the equation above, taking logarithms of both sides we see that lt = -ln( N/ N 0) = ln( N 0/ N), so our equation for t isįor convenience, we'll now write F for N/ N 0. Suppose that we invert our function for N/ N 0 in terms of t, to get an expression for t as a function of N/ N 0. This question can be answered using a little bit of calculus. On average, how much time will pass before a radioactive atom decays? The steps are the same as in the case of photon survival. 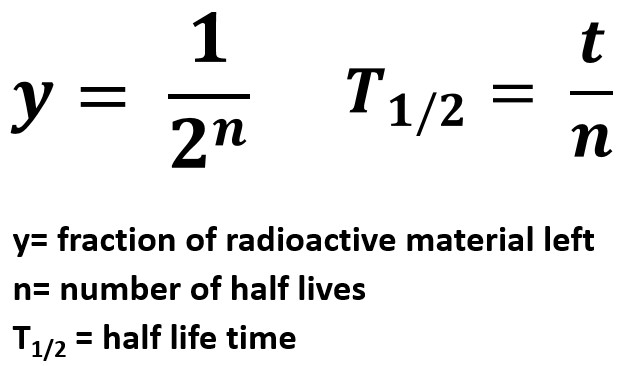
We can easily find an expression for the chance that a radioactive atom will "survive" (be an original element atom) to at least a time t. Where the quantity l, known as the "radioactive decay constant", depends on the particular radioactive substance.Īgain, we find a "chance" process being described by an exponential decay law. We get an expression for the number of atoms remaining, N, as a proportion of the number of atoms N 0 at time 0, in terms of time, t: The same solution that we saw in the case of light attenuation. We end up with a solution known as the "Law of Radioactive Decay", which mathematically is merely If we have a sample of atoms, and we consider a time interval short enough that the population of atoms hasn't changed significantly through decay, then the proportion of atoms decaying in our short time interval will be proportional to the length of the interval. However, now the "thin slice" is an interval of time, and the dependent variable is the number of radioactive atoms present, N( t). To show this, we needed to make one critical assumption: that for a thin enough slice of matter, the proportion of light getting through the slice was proportional to the thickness of the slice.Įxactly the same treatment can be applied to radioactive decay. In the previous article, we saw that light attenuation obeys an exponential law. Law, and explains how this information allows us to carbon-date artefacts such as the Dead Sea Scrolls. In this second article he describes the phenomenon of radioactive decay, which also obeys an exponential The following equation gives the quantitative relationship between the original number of nuclei present at time zero (N_0\) and the number (\(N\)) at a later time \(t\).In his article Light Attenuation and Exponential Laws in the last issue of Plus, Ian Garbett discussed the phenomenon of light attenuation, one of the many physical phenomena in which the exponential function crops up. It is also applicable to the decay of excited states in atoms and nuclei. The concept of half-life is applicable to other subatomic particles, as will be discussed in Particle Physics. Nuclides with the shortest half-lives are those for which the nuclear forces are least attractive, an indication of the extent to which the nuclear force can depend on the particular combination of neutrons and protons. \)y for the least unstable, or about 46 orders of magnitude. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
